As winner of numerous international awards, Bruschi Spa is known for its innovative approach in design and technology. We are glad to share our insights and experiences with the industry members.
Die casting surface treatments: avoiding blisters, pitting and other defects in plating
In this post we are going to list the most common defects in zinc die casting surface treatments and explain how to prevent them. We are going to analyze the following defects: blisters, pitting, burnings, nodules and flaking.

Surface finishing has a crucial role in zinc alloy components production: often the main reason to choose Zamak for a component is the wide range of finishing and colorings available, either through plating or painting. For this reason it is important to know and learn to prevent defects linked to the final surface quality of the diecast.
Die casting surface treatments defects can be linked to many different causes, ranging from the quality of the casting to issues in pre-finishing or finishing phases. The term pre-finishing includes all the operations necessary to prepare the product for following treatments of electrodeposition or painting, such as deburring, vibrating, cleaning and polishing. Depending on the root cause, the kind of defect may vary, but some are more common than others.
Among the most widespread defects in die casting surface treatments, we find blisters and pitting: they can be caused by a number of factors, therefore it may be necessary a more in-depth analysis to identify the cause and proceed to correction.
In addition to blisters and pitting, there are three types of surface defects easily recognizable: burnings, nodules and flaking.
Blisters
Blisters look like small bulges on the surface of the components and their dimension can vary depending on the cause of their formation. They can be caused by errors in casting, pre-finishing phases or during electrodeposition.
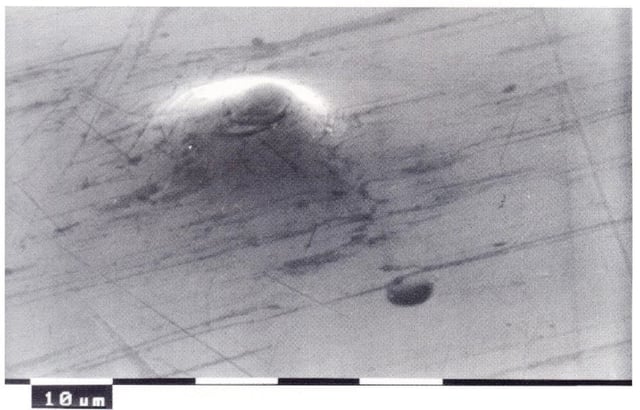
Casting blisters
This kind of blisters can be caused by contaminants in the alloy, such as lead and cadmium, or by intergranular corrosion, but they can also be linked to defects in casting such as cold laps and surface porosity.
In this case, blisters are round-shaped and big, and they feel hard to the touch. For a better analysis it will be necessary to cut the blister with a sharp blade: it will be then possible to observe that, by sectioning the blister, the diecast will be cut too. These traits identify blisters formed due to underlying porosity in die casting phase: in order to solve this issue, it will be necessary to reduce surface porosity.
Pre-finishing blisters
As stated above, not all blisters are caused by casting defects: some can form due to errors in pre-finishing phases. For example, during the preparation of a piece for electrodeposition, special attention should be dedicated to the detection of oxidation, corrosion and other superficial contaminants. If the surface isn’t perfectly clean, groups of round blisters will form after electrodeposition: by cutting through these blisters, the underlying metal will be left exposed and, observing its section at a microscope, it would be easy to spot the oxidated layer that caused the defect.
In order to avoid these situations, it is advisable to prevent delays between casting, cleaning and electrodeposition so as to reduce the chance of surface oxidation. When this is not possible, for example when relaying on an external supplier, another way to prevent blistering is to apply a corrosion inhibitive solution to the components and remove it right before plating. If, despite taking all the mentioned precautions, traces of oxidation are found on the surface, it is possible to remove it through mechanical vibrating or cleaning it with acids.
Blisters caused by electrodeposition
Another easily recognizable kind of blister is the one caused by a lack of adherence between nickel and copper layers, or between nickel and chrome. These blisters often pop up in large groups, have an irregular shape and can be found on the whole surface of the component, sometimes far from one another. Unlike casting blisters, they are soft and squishy and, after removal, the underlying copper or nickel coating will be visible.
The formation of these blisters can be prevented by making sure that the copper layer is sufficiently thick, by leaving the shortest possible time lapse between nickeling and copper plating and by regularly checking baths for contaminants.
Pitting
Unlike blisters, pitting is always caused by mistakes during pre-finishing or finishing phases. They look like small lumps whose shape and position can vary depending on the root cause. There are 4 main causes of pitting: solvents or corrosive liquids on the surface, gas trappings and hydrogen bubbles in the galvanic bath.

When pitting is due to solvent residues that prevent the correct adherence of copper layer, lumps will be disposed in circle or chains, whereas when the cause is an unexpected splash of corrosive liquid, pitting will be distributed in an irregular shape.
In the first case, to solve the problem it is possible either to modify the degreasing system, or to add a cooling bath between degreasing phase and plating phase. Temperature plays a crucial role in these phases: sometimes it can be enough to lower the temperature of the casting or to wait longer between liquid and steaming treatments to prevent the formation of pitting.
Instead, when pitting appears in irregular shapes, the solution can be to control the source of splashes and fix it, or to apply a thicker copper layer so as to cover the defect.
Sometimes pitting can be randomly disposed and look like small craters with high edges: this happens because during the nickel-plating phase some hydrogen bubbles in the galvanic bath stick to component’s surface. The presence of these bubbles can be caused by different factors:
- Inadequate mixing of galvanic bath
- Low level of surfactants
- Oily layer on the surface of nickeling bath
- Dust particles sticking to the piece’s surface
To prevent the formation of this defect, it will be necessary to monitor the amount of surfactants and the mixing of the galvanic bath, in addition to protecting the surroundings to prevent infiltration of powders.
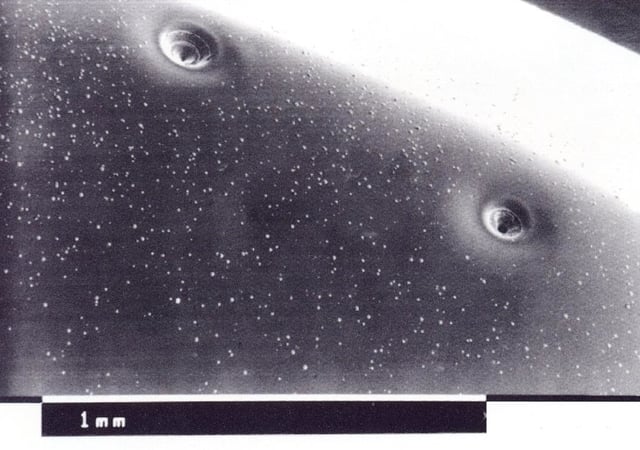
One last kind of pitting is caused by the presence of gas and bubbles in the galvanic bath: bubbles stick to the surface of the component, creating round and deep pitting, close to each other and covering the whole surface. To prevent the formation of these defects, it will be necessary to spot the gas source and to check baths’ state.
Sometimes the solution itself is naturally rich in gas, which gets released as soon as the mixture reaches a certain temperature. In times like these it is possible to remove gas by overheating the solution, bringing it to an higher temperature than the one requested for galvanization process and cooling it down again before using it.
Burnings
They look like a brown spot and a burnt crown, and are caused by a high pitch in current density or by a contamination of the bath: this contamination can derive from the use of the solution, because during the plating process some zinc particles may detach from the component and alter the solution.
To avoid these formations is then sufficient to periodically change the bath mix or to dilute the solution by adding more electrolytes.
Nodules
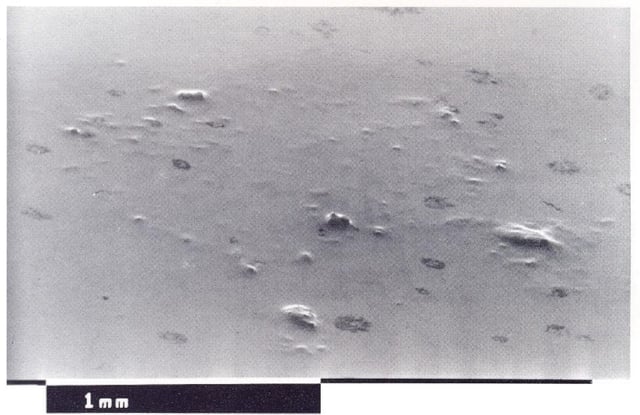
Nodules look like small bumps that make the surface of the casting grainy: they derive from particulate matter depositing on the piece or in the nickel-plating tank. To avoid these formations it is advisable to periodically filter galvanic mixture and to be careful when cleaning the pieces.
Flaking
Flaking is caused by a faulty attachment of the external nickel layer: it looks like compact and long vines, with different shapes and dimensions. They are easily scratched with nails or a blade, and when removed, they leave the underlying coating on display.
The root causes of these defects are identifiable thanks to their position: if detaching is formed between copper and nickel coatings, it may be caused by contaminated solution, delays in the process or insufficient activation. Flaking can also be formed between polished layer and semilucid nickel layer, and their cause is to be found in the passivation of the semilucid coating, whereas if the formation is located inside the polished nickel layer, it is usually due to flicks or current interruptions during the plating.
Generally, to avoid the creation of flaking it is necessary to keep the solution pure, or to monitor the current flow to avoid interruptions during the process.
To sum up, we have seen how working with chroming and electrodeposition on zinc alloy products can induce many surface defects that can be linked to casting defects or processing errors. Some can however be avoided through constant monitoring of galvanic baths and solutions, and a careful cleaning of the diecast and the working environment.
To learn more about zinc die casting surface treatments and how to fix common defects in die casting, subscribe to our blog or contact us for a tailor-made solution using the form at the end of the page.

